Earlier studies indicated that cell membranes present a barrier to the free passage of hydrophilic biopolymers like polysaccharides. They can initiate intracellular signaling cascades and control cellular physiological functions, like activating immune responses, through their binding to pattern recognition receptors (PRRs) and various other target molecules, including scavenger receptors (SRs), Toll-like receptors (TLRs), complement receptor 3 (CR3, αMβ2 integrin, CD11b/CD18), C-type lectin receptors (CLRs), and mannose receptors (MRs).
This blog post introduces research summarizing key findings on the molecular targets of fucoidan and provides a detailed description of its pharmacological mechanisms from the perspective of receptors and affinity molecules.
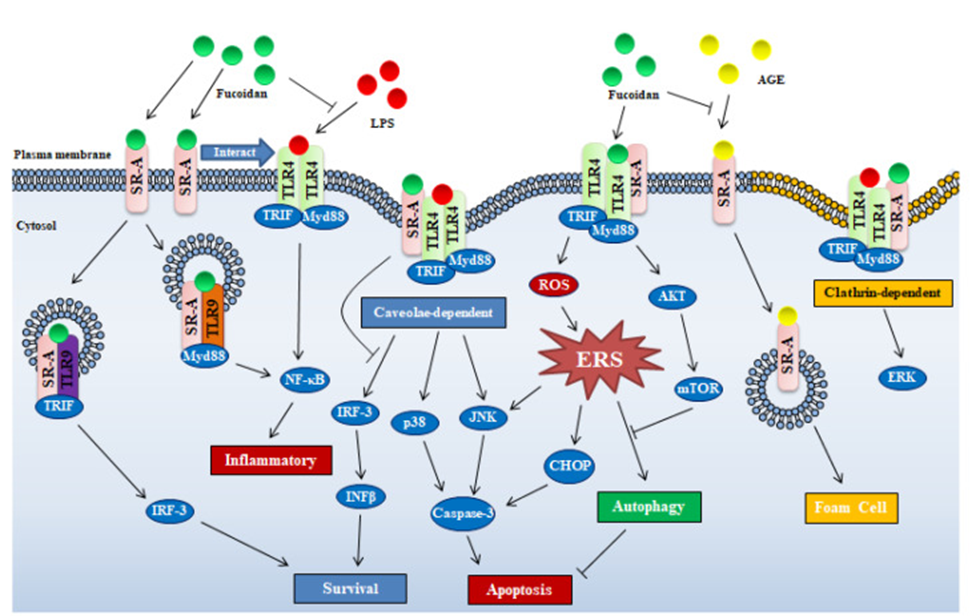
Fucoidan attaches to the sites on macrophages that recognize low-density lipoprotein (LDL). These sites are the class A scavenger receptors (SR-A), which are more prevalent in macrophages and play roles in managing lipid levels, the development of atherosclerosis, and various metabolic functions. SRs recognize and internalize various macromolecules and polyanionic compounds, including LDL, lipopolysaccharide (LPS), and lipoteichoic acid (LTA). SR-A is a widely distributed membrane receptor and has been found in many cell types, including macrophages, renal tubular epithelial cells, mesenchymal cells, dendritic cells, endometrial cells, vascular smooth muscle cells, and endothelial cells.
This suggests that fucoidan exerts a wide range of biological functions in various organs/tissues through binding to SR-A. It’s important to note that the majority of research papers concentrate on macrophages. A common theory among researchers is that SR-A’s intracellular signaling arises from its synergistic interplay with other membrane receptors, such as TLRs. Seimon et al. found that fucoidan/SR-A, in cooperation with TLR4, induces JNK-dependent apoptosis in the endoplasmic reticulum of stressed peritoneal macrophages, shifting TLR4 signaling from pro-survival to pro-apoptotic. Also, Yu et al. demonstrated that SR-A is required for LPS-induced TLR4-mediated NF-κB activation in fucoidan-exposed macrophages, although SR-A was not involved in NF-κB gene expression. As depicted in Figure 1, these findings indicate that for intracellular signaling to occur, fucoidan/SR-A requires a cooperative interaction with TLR4, even though SR-A binds fucoidan with high affinity.
Fucoidan is recognized as being primarily taken into cells via SR-A, an endocytosis receptor. TLR4 is a receptor that is activated by fucoidan, triggering an inflammatory response through the NF-κB signaling pathway. However, the ultimate effect of fucoidan is widely recognized as anti-inflammatory. The anti-inflammatory actions of fucoidan, notably when LPS or similar inflammatory compounds are involved, are understood to be a result of SR-A’s absorption of fucoidan, the clearance of TLR4 ligands, and the subsequent suppression of TLR4 activation. Pei et al. showed that in rat microglia HAPI cells, fucoidan significantly inhibited LPS uptake but did not inhibit LPS-induced superoxide production. The data implies that fucoidan’s interaction with SR-A led to the inhibition of LPS uptake. Similarly, fucoidan suppressed SR-A-mediated uptake of polyanionic polypeptides into J774 cells. Advanced glycation end products (AGEs), which are produced when proteins are exposed to sugars for extended periods, are another class of ligands for SR-A. The affinity of fucoidan for SR-A suggests its potential as an inhibitor of toxic AGE uptake, offering a means to mitigate the toxicity of AGE endocytosis (see Figure 1).
Two endocytic mechanisms are employed by macrophages for the uptake of the SR-A/ligand complex: clathrin-dependent and caveolae-dependent pathways. The latter pathway is necessary for apoptosis induction by SR-A/fucoidan uptake, and this is associated with the activation of p38 kinase and c-Jun N-terminal kinase (JNK) (see Figure 1). Similarly, under the action of SR-A ligands, SR-A interacts with major scaffold proteins and activates the JNK signaling pathway via the caveolin-mediated endocytosis pathway.
A crucial factor in inducing macrophage apoptosis, as illustrated in Figure 1, is the inhibition of endoplasmic reticulum stress (ERS)-induced autophagy in macrophages through the fucoidan/SR-A-mediated activation of the AKT/mTOR/p70S6K pathway. Seimon et al. speculated that the induction of apoptosis in macrophages with ERS by fucoidan via the SR-A receptor requires coordination with the TLR4 receptor.
According to existing research, fucoidan’s impact on cellular functions through SR-A can be understood in two ways: the first involves fucoidan/SR-A facilitating intracellular signaling by engaging with other receptors and pathways, such as ER stress and endocytosis. Fucoidan also operates by competing for SR-A binding, which effectively neutralizes the influence of other ligands on cellular activity (see Figure 1). Despite the existence of many mechanisms, it is clear that SR-A plays a crucial role in the regulation of immune function by fucoidan. Furthermore, although most studies on SR-A and fucoidan focus on macrophages in vitro, SR-A has also been reported to be expressed in vascular smooth muscle cells, endothelial cells, human lung epithelial cells, microglia, astrocytes, and mouse embryonic fibroblasts. Considering the expression distribution of SR-A, it is understood that fucoidan exerts its function not only in immune cells but also in many parts of the body. Due to fucoidan’s intricate regulation of intracellular signals through SR-A, pinpointing its exact function in diseases solely from in vitro studies on specific cell types presents a significant challenge.
Toll-like receptors (TLRs) are a group of evolutionarily conserved transmembrane proteins that recognize endogenous and exogenous ligands. TLRs have a leucine-rich repeat at the N-terminus and a Toll/IL-1R homologous domain at the C-terminus, and they transmit signals downstream via adapter proteins such as MyD88 (myeloid differentiation factor), MyD88 adapter-like protein, TIR domain-containing adapter-inducing IFN-β (TRIF), and TRIF-related adapter molecule. To date, a total of 13 types of TLRs have been identified, including TLR1-TLR11 expressed in humans.
Innate immune receptors TLR2 and TLR4 recognize naturally occurring polysaccharides. This recognition triggers NF-κB signaling, leading to the rapid movement of NF-κB from the cytoplasm to the nucleus. This translocation then controls the expression of specific genes, which in turn encourages the release of inflammatory cytokines and influences immune responses. According to Teruya et al., fucoidan is identified by a complex comprising TLR4, SR-A, and CD-14. Subsequently, it activates the MAPK signaling pathway, which results in the induction of NO production within macrophages while p38 MAPK, SAPK/JNK, and ERK1/2, which are part of the MAPK family, are downstream signaling pathways activated by fucoidan through TLR4, ERK1/2 is not implicated in the NO induction process within macrophages. Fucoidan derived from brown algae activates NF-κB via TLR-2 and TLR-4 on HEK293 cells, and these TLRs showed different affinities for fucoidans from different sources. TLR4 activated by fucoidan induces ROS-related endoplasmic reticulum stress (ERS) by activating the PERK–ATF4–CHOP pathway, and promotes caspase-3 activation via the TLR4–ERS–CHOP pathway, leading to apoptosis in lung cancer cells (see Figure 1).
While it is reasonable that TLR activation mediates the synthesis and secretion of inflammatory cytokines, fucoidan is also well-known to have anti-inflammatory effects. Studies have revealed that SR-A has a negative regulatory effect on immune cell activation mediated by TLR4. Therefore, the interplay between SR-A and TLR4 is considered important for fucoidan-related biological functions.
Additionally, fucoidan’s activity also involves TLR3 and TLR9. The interaction of internalized SR-A with TLR3 promotes cell survival via the TLR3/IRF-3 pathway, while its interaction with TLR9 mediates cell inflammation or death through the TLR9/TRIF/NF-κB/TNF-α pathway. This implies that fucoidan influences cell survival or triggers cell death by facilitating the interaction between SR-A and TLR3/TLR9, as illustrated in Figure 1.
C-type lectin receptors (CLRs), members of the membrane-bound C-type lectin (CLEC) family, are another group of pattern recognition receptors (PRRs). CLRs not only recognize various antigens but also participate in intercellular signaling, aiding in the induction of innate immunity by macrophages and dendritic cells. These receptors can recognize fungal cell wall components such as β-glucan, mannose, and fucoidan. Fucoidan inhibits cell adhesion by binding to the Ly49 family, a member of the CLR family. Additionally, fucoidan interacts with the osteoclast inhibitory lectin (OCIL) family, a type of CLR found on NK cells; however, this interaction doesn’t alter OCIL’s ability to prevent osteoclast development.
Cardlan, a high-molecular-weight glucan, activates both human and mouse Dectin-1, while low-molecular-weight glucans activate only cells expressing human Dectin-1, but not those expressing mouse Dectin-1. It is believed that this is caused by variations in the amino acid makeup of the intracellular sections of human and mouse Dectin-1, resulting in differing receptor sensitivities.
The C-type lectin 2 (CLEC-2) superfamily, a transmembrane receptor primarily expressed on platelets, is activated when a phosphorylated atypical immunoreceptor tyrosine-based activation motif binds to the tandem Src homology 2 domain of spleen tyrosine kinase, promoting platelet aggregation. Fucoidan has been identified as an activator of this receptor, which in turn promotes platelet aggregation. This process occurs through a signaling pathway involving tyrosine kinases, and it has been observed to decrease bleeding time in animal models of hemophilia. Fucoidan-induced activation of CLEC-2 triggers phosphorylation of PLCγ2 in the signalosome, leading to the production of sufficient amounts of IP3 to induce transient fluctuations in intracellular Ca2+ concentration (calcium oscillations), resulting in platelet aggregation. Glycoprotein VI (GPVI), another platelet surface receptor, is also activated by fucoidan, which subsequently augments platelet aggregation. Martyanov et al. believe that the procoagulant effect associated with IP3 outweighs the anticoagulant effect of fucoidan. For these reasons, fucoidan should not be employed instead of low molecular weight heparin.
Additionally, fucoidan’s diverse biological effects are mediated by numerous key proteins, both within and outside of membranes. These target molecules include integrins (CR3 and αVβ3), VEGF, CXCL12/CXCR4, elastin peptide receptors, TGF-β1, and certain extracellular matrix (ECM) proteins. This research aims to enhance our comprehension of fucoidan’s pharmacological actions, identify novel applications, and facilitate the creation of associated clinical drugs, dietary supplements, and health-focused foods.
Source: Mar Drugs. 2020 Jul 22;18(8):376. doi: 10.3390/md18080376
