Diabetes, a long-term metabolic condition defined by elevated blood glucose levels from inadequate insulin, poses significant health risks, such as the development of diabetic foot ulcers (DFUs). These ulcers are often exacerbated by polymicrobial infections involving bacteria such as Staphylococcus aureus and Acinetobacter baumannii. These pathogens are known for their antibiotic resistance, making treatment difficult. This blog post introduces research by Mohsen Nazari et al., titled “The antimicrobial and antibiofilm effects of gentamicin, imipenem, and fucoidan combinations against dual-species biofilms of Staphylococcus aureus and Acinetobacter baumannii isolated from diabetic foot ulcers.” Researchers examined the combined impact of gentamicin, imipenem, and fucoidan, which is an antimicrobial sulfated polysaccharide derived from the brown seaweed Sargassum angustifolium, on planktonic and biofilm cultures of Staphylococcus aureus and Acinetobacter baumannii.
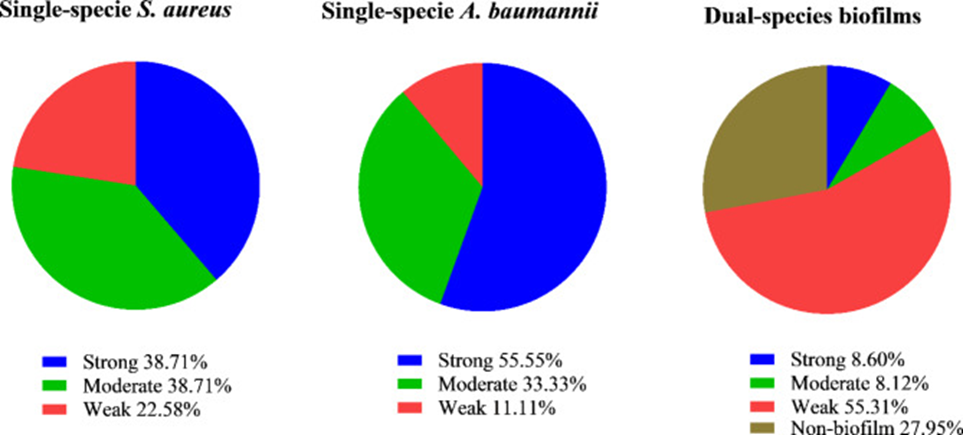
The first step involved antibiotic susceptibility testing and molecular biological analysis (targeting the FOX disk and mecA gene), which determined that 80.6% (25 isolates) of Staphylococcus aureus strains presented characteristics aligning with methicillin-resistant Staphylococcus aureus (MRSA). Variability in biofilm formation ability was observed in both Staphylococcus aureus and Acinetobacter baumannii isolates, with OD values ranging from 0.2 to 2.8 for all isolates.
Antimicrobial resistance rates were evaluated using disk diffusion data from clinical isolates. The resistance rates of Staphylococcus aureus (S. aureus) to IMP, TS, E, CD, GM, LZD, and VAN were 80.6%, 67.7%, 64.5%, 58.5%, 51.6%, 0%, and 0%, respectively. Similarly, based on disk diffusion data, the antimicrobial resistance rates of Acinetobacter baumannii (A. baumannii) clinical isolates were PIP 100%, CEF 100%, AM 100%, TET 92.6%, AUG 85.1%, GM 77.7%, MER 77.7%, CIP 74.0%, and IMP 55.5%. Overall, 77.4% of S. aureus isolates and 100% of A. baumannii isolates exhibited multidrug resistance (MDR). A significant correlation has been observed, according to the results, between antimicrobial resistance and the formation of substantial biofilms in clinical isolates of S. aureus.
Next, they investigated various conditions for all isolates (31 S.aureus and 27 A. baumanii), revealing that 72 conditions formed strong dual-species biofilms, while 67 formed moderate dual-species biofilms out of a total of 837 conditions. Detailed information for all isolates is presented in Figure 1. In some cases, although OD was higher than that of the negative control, growth of both bacteria after biofilm formation was not observed in culture on MSA and Leeds media, in which case it was reported as non-biofilm formation.
The geometric MIC values for Staphylococcus aureus cultures grown in isolation were 12.12 µg/mL for gentamicin, 19.84 µg/mL for imipenem, and 48.50 µg/mL for fucoidan. For isolated Acinetobacter baumannii cultures, these values were 22.62 µg/mL for gentamicin, 9.18 µg/mL for imipenem, and 31.20 µg/mL for fucoidan. Similarly, the geometric mean MBC values for gentamicin, imipenem, and fucoidan in Staphylococcus aureus were 119.42, 48.50, and 88.37 µg/mL, respectively, and in Acinetobacter baumannii, they were 128, 14.92, and 62.62 µg/mL, respectively. Furthermore, the geometric mean MBC/MIC ratios for gentamicin, imipenem, and fucoidan in Staphylococcus aureus were 9.84, 2.46, and 2.14, respectively, and in Acinetobacter baumannii, they were 5.65, 1.74, and 2, respectively.
Under co-culture conditions, six isolates—S. aureus 6, S. aureus 7, S. aureus 22, A. baumannii 1, A. baumannii 8, and A. baumannii 20 were selected for study. These isolates were capable of forming mixed-species biofilms under nine different conditions (3×3). Of these conditions, strong mixed-species biofilms were formed under eight conditions, and a moderate mixed-species biofilm was formed under one condition. MIC and MBC values were evaluated under co-culture conditions where growth inhibition or killing of both bacterial species was observed (see Figure 2A). The geometric mean MIC values, MBC values, and MBC/MIC ratios for gentamicin, imipenem, and fucoidan were 5.87 µg/mL, 11.75 µg/mL, and 1.85 for gentamicin; 3.70 µg/mL, 8 µg/mL, and 2.16 for imipenem; and 19.65 µg/mL, 42.48 µg/mL, and 2.16 for fucoidan, respectively.
The geometric mean MBIC values for isolated Staphylococcus aureus strains were 55.71 µg/mL for gentamicin, 68.59 /mL for imipenem, and 153.89 µg/mL for fucoidan. For isolated Acinetobacter baumannii strains, the respective values were 119.42 µg/mL for gentamicin, 48.50 µg/mL for imipenem, and 101.49 µg/mL for fucoidan.
Furthermore, the geometric mean MBEC values for Staphylococcus aureus were 315.17 µg/mL for gentamicin, 207.93 µg/mL for imipenem, and 353.55 µg/mL for fucoidan. For Acinetobacter baumannii, the respective values were 362.03 µg/mL for gentamicin, 157.58 µg/mL for imipenem, and 189.46 µg/mL for fucoidan.
For the mixed-species biofilm, the geometric mean MBIC and MBEC values were 18.66 µg/mL and 59.25 µg/mL for gentamicin, 25.39 µg/mL and 74.65 µg/mL for imipenem, and 53.53 µg/mL and 99.19 µg/mL for fucoidan, respectively. Details of these results are shown in Figure 2B.
This research identified the geometric mean concentrations that demonstrated the most significant synergistic effects when using various antibiotic combinations to treat Staphylococcus aureus and Acinetobacter baumannii in dual-species biofilms. Using FBICi, the combination of gentamicin and imipenem yielded values of 0.82, 0.59, and 0.29 for the three selected isolates, respectively. The combination of gentamicin and fucoidan yielded values of 0.47, 0.44, and 0.28, respectively. Similarly, the combination of imipenem and fucoidan yielded values of 0.47, 0.46, and 0.37.
The concentrations demonstrating ideal synergistic effects were subsequently assessed via FBECi. For the combination of gentamicin and imipenem, the geometric mean values were 0.98, 0.69, and 0.24. For the combination of gentamicin and fucoidan, the values were 0.55, 0.58, and 0.36. Finally, for the combination of imipenem and fucoidan, values of 0.52, 0.48, and 0.39 were obtained for the same isolates.
The synergistic potential of gentamicin, imipenem, and fucoidan in eradicating or neutralizing dual-species biofilms of S. aureus and A. baumannii was evaluated using FE-SEM. The study indicated that when fucoidan and antibiotics were combined at their FBIC levels, they substantially broke down biofilm structures. Specifically, the synergy between fucoidan and the antibiotics at these concentrations led to substantial disruption of the dual-species biofilm, causing bacterial lysis and a marked reduction in the overall biofilm biomass. The organized biofilm was disrupted, leading to the formation of smaller, less cohesive units. Moreover, the combination of gentamicin and imipenem also showed a strong synergistic effect, leading to the reduction of large biofilm biomass, bacterial lysis, and the formation of detached bacterial cells, see Figure 3.
The expression levels of the icaA gene in Staphylococcus aureus and the bap gene in Acinetobacter baumannii were evaluated after 24-hour exposure to combinations of gentamicin and imipenem, gentamicin and fucoidan, and imipenem and fucoidan, each at 1/2 MBIC concentration. In single-species biofilms, icaA expression was downregulated by 2.98–1.52 times, and bap expression was downregulated by 3.15–1.94 times. In mixed-species biofilms, S. aureus icaA expression decreased by 5.92–3.64 times, and A. baumannii bap expression decreased by 5.78–4.17 times. When gentamicin and fucoidan were combined, icaA expression was downregulated by 3.32–1.90 times and bap expression by 2.86–1.79 times in single-species biofilms. In mixed-species biofilms, S. aureus icaA expression decreased by 6.12–4.20 times, and A. baumannii bap expression decreased by 5.68–3.79 times. When imipenem and fucoidan were combined, icaA expression was downregulated by 3.1–1.72 times and bap expression by 3.12–1.68 times in single-species biofilms. In mixed-species biofilms, S. aureus icaA expression decreased by 5.53–3.75 times, and A. baumannii bap expression decreased by 6.87–4.48 times. ANOVA analysis revealed a significant difference in gene expression between single-species and mixed-species biofilms.
The cytotoxicity assessment showed that HSF-PI 16 cells exhibited 78.3%, 42.1%, and 11% cytotoxicity at fucoidan concentrations of 1000, 500, and 250 µg, respectively. Importantly, when fucoidan was combined with the antibiotics imipenem (32 and 64 µg) and gentamicin (32 and 64 µg) at concentrations of 125, 62.5, 32.1, 15.6, and 7.8 µg, which showed optimal synergistic effects, no cytotoxic effects were detected in HSF-PI 16 cells.
Research into the combined use of gentamicin, imipenem, and fucoidan has shown that these substances work together effectively against mixed-species biofilms formed by Staphylococcus aureus and Acinetobacter baumannii, suggesting a promising avenue for better treatment results. By leveraging the synergistic effects of these antibiotics, it may be possible to improve treatment outcomes for mixed-species biofilm infections. According to researchers, introducing bacteria that compete with the existing ones in single-species biofilms could lead to more effective antimicrobial treatments.
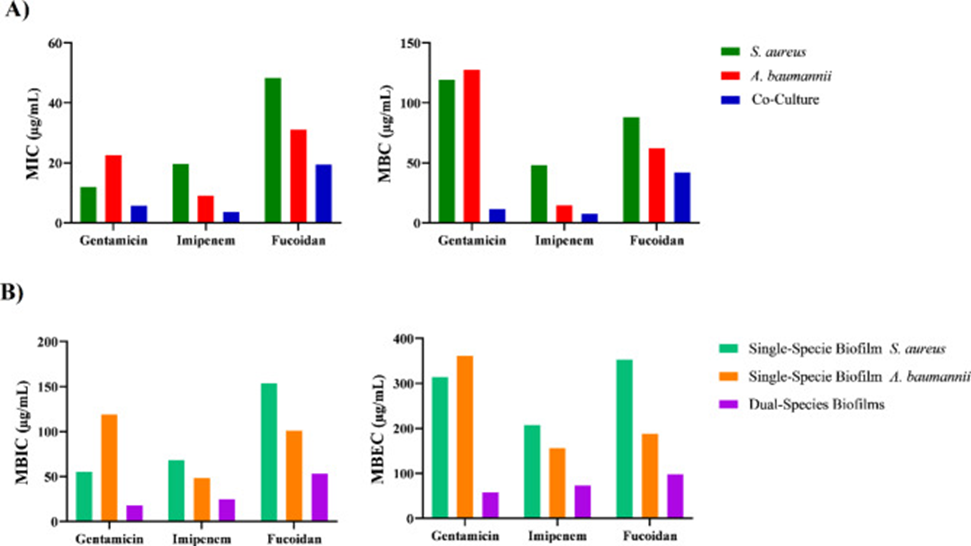
2B) The geometric mean values of the minimum biofilm inhibitory concentration (MBIC) and minimum biofilm eradication concentration (MBEC) are depicted for S. aureus and A. baumannii in both single-species biofilms and in dual-species biofilms, where both bacteria are present together
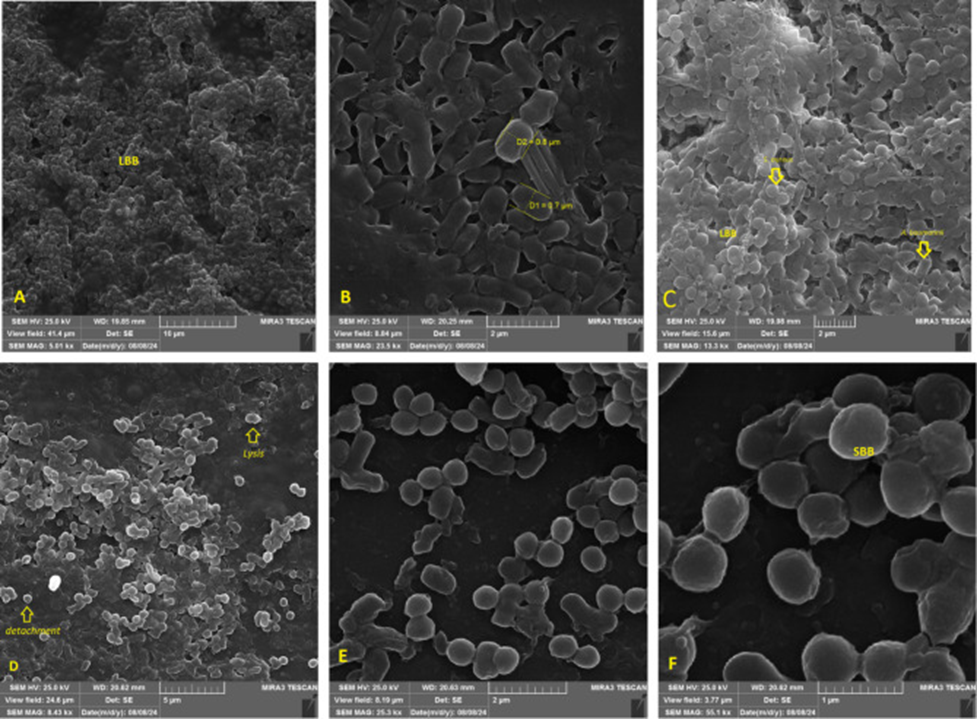
Source: Ann Clin Microbiol Antimicrob. 2024 Nov 15;23:101. doi: 10.1186/s12941-024-00760-w
