The bone marrow serves as the site where stem cells differentiate and mature into white blood cells, playing a vital role in the body’s defense mechanisms. Leukemia arises when faulty chromosomes lead to a loss of control over cell division. Consequently, cells with these chromosomal defects proliferate uncontrollably or avoid apoptosis, resulting in leukemia. The research featured in this blog is titled “Induction of Apoptosis by Fucoidan in Human Leukemia U937 Cells through Activation of p38 MAPK and Modulation of Bcl-2 Family,” and it was conducted by Hyun Soo Park et al.
Fucoidan, a complex sugar molecule derived from brown seaweed, exhibits diverse biological properties such as antiviral, antimicrobial, and anti-inflammatory actions. An MTT assay was employed to examine how fucoidan affects leukemia cell proliferation by measuring cell viability. The viability of U937 cells decreased with fucoidan treatment, and this reduction was linked to both the concentration of fucoidan and the duration of exposure.
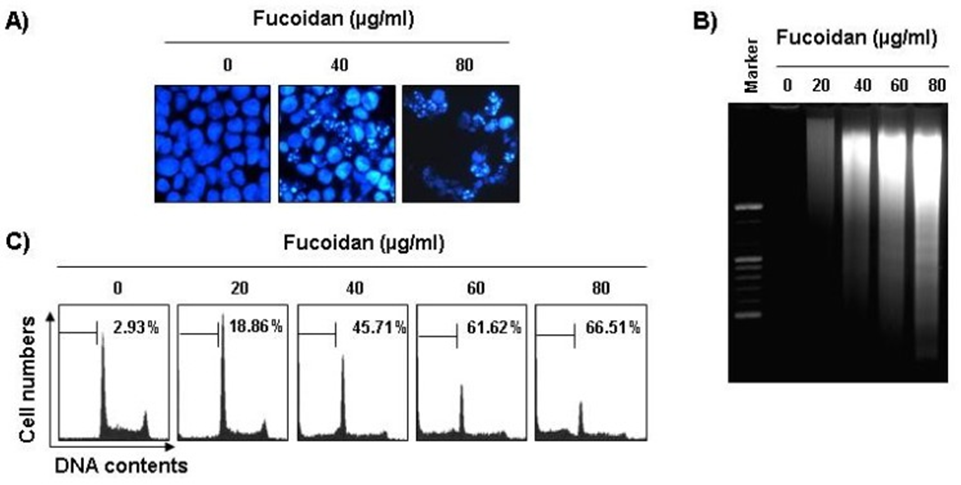
Following this, experiments were carried out to investigate whether fucoidan’s inhibitory effect on cell viability was a consequence of apoptosis. Morphological examination of apoptosis was conducted by staining nuclei from untreated and fucoidan-treated cells with 4,6-diamidino-2-phenylindole (DAPI) solution and subsequently observing them. The nuclear structure of control cells remained unaltered, in contrast to fucoidan-treated U937 cells, which exhibited apoptotic features like chromatin condensation and nuclear fragmentation, shown in Figure 1A. The formation of nucleosome DNA ladders was observed in U937 cells treated with fucoidan at concentrations greater than 40 μg/mL for 48 hours, according to agarose gel electrophoresis results (see Figure 1B). Additionally, cell cycle analysis was used to measure the amount of apoptotic cell death. In U937 cells, as depicted in Figure 1C, fucoidan treatment led to a significant rise in apoptotic sub-G1 phase cells, a dose-dependent phenomenon.
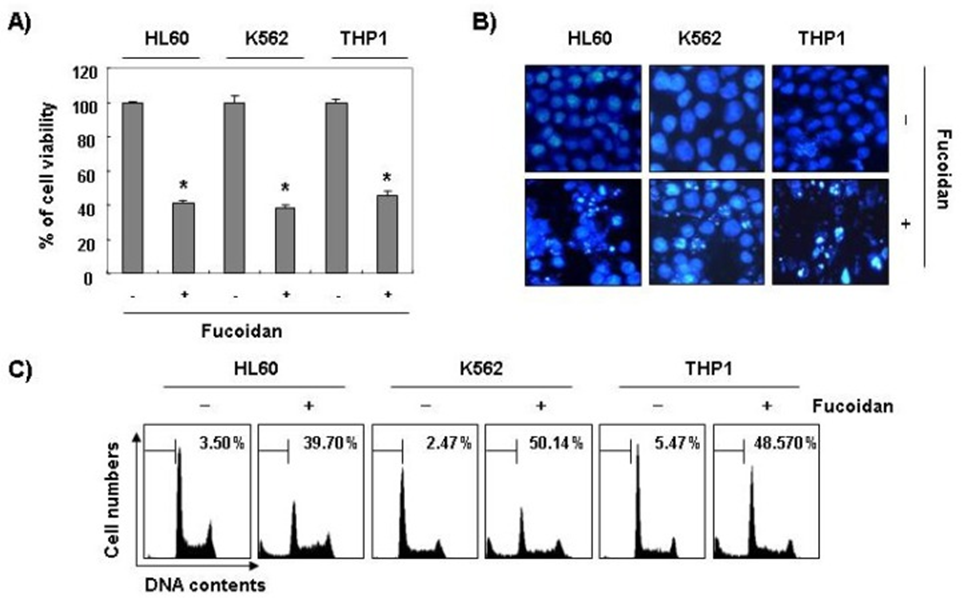
Beyond this, the research indicated that fucoidan played a significant role in diminishing the survival rate of cells and initiating the process of apoptosis in various other types of leukemia cells, such as HL60, K562, and THP1, which is further illustrated in Figure 2. These findings indicate a link between the growth inhibition seen with fucoidan and the triggering of apoptosis in leukemia cells.
Apoptosis, a programmed cell death process, is significantly influenced by caspases in both its intrinsic and extrinsic pathways. These enzymes contribute to common forms of apoptosis by breaking down various cellular components, including PARP. To better understand how fucoidan triggers apoptosis, the researchers investigated its impact on caspase protein levels and activity, along with their inhibitors, the inhibitor of apoptosis (IAP) family proteins. The Western blot analysis demonstrated that fucoidan exposure enhanced the levels and activity of active caspases 3, 8, and 9, with the effect increasing with concentration. Following Western blot analysis, it was observed that U937 cells treated with fucoidan exhibited ongoing proteolytic cleavage of PARP. Additionally, an 85-kDa protein, which is a downstream target of activated caspase 3, accumulated in these cells.
To show that caspase activation is a crucial part of the fucoidan-induced apoptosis process, U937 cells were first treated for an hour with a broad-spectrum caspase inhibitor (z-VAD-fmk) and another caspase inhibitor that might be specific, then exposed to fucoidan for 48 hours. Pretreatment with the caspase inhibitor significantly inhibited the expansion of the sub-G1 cell population and reversed the fucoidan-induced loss of viability. The data presented in this shows that U937 cells undergo apoptosis as a consequence of fucoidan treatment, with this cellular demise being facilitated by a caspase-dependent signaling pathway. Additionally, fucoidan therapy decreased the amounts of IAP family proteins, including XIAP and cIAP-1. The proteins in question are known to interact with caspases, leading to their inactivation. The findings indicate that fucoidan treatment triggers apoptosis in U937 cells by activating caspases and decreasing IAP family proteins.
The research sought to determine if the mitochondrial pathway plays a role in fucoidan-induced apoptosis in U937 cells. MMP and Bcl-2 family protein expression was quantified through a procedure that incorporated flow cytometry with a fluorescent cationic dye and Western blot analysis. Fucoidan treatment led to a decrease in MMP that was proportional to its concentration when compared to untreated controls. Fucoidan also lowered the intact pro-apoptotic protein Bid, while the truncated form remained undetected. In addition, the amount of Bcl-xL protein in U937 cells was lowered by fucoidan treatment, with the effect increasing with dosage. Meanwhile, the levels of Bcl-2, another anti-apoptotic protein, and Bax, a pro-apoptotic protein, both crucial for mitochondrial stability, showed slight increases and decreases, respectively, following fucoidan exposure.
The Bcl-2 protein typically attaches to Bax, a pro-apoptotic protein found in mitochondria, thereby preventing programmed cell death. This led the researchers to investigate further, using Co-IP assays and mitochondrial fractionation, if Bax relocated from the cytosol to mitochondria after fucoidan administration. Fucoidan treatment significantly increased the interactions between Bax and Bcl-2 and between Bax and Bcl-xL. In particular, the binding between Bax and Bcl-2 was found to be stronger than that between Bax and Bcl-xL. The Fucoidan treatment led to a reduction in cytoplasmic Bax levels and a simultaneous increase in cytoplasmic cytochrome c. The concentration of Bax in mitochondria increased significantly, while the concentration in mitochondria decreased significantly. The research suggests that fucoidan prompts Bax to migrate from the cytoplasm to the mitochondria. This migration enhances the association between Bax and Bcl-2, diminishes MMP, and ultimately causes mitochondrial impairment. The impaired mitochondria then release cytochrome c into the cytoplasm, initiating apoptosis.
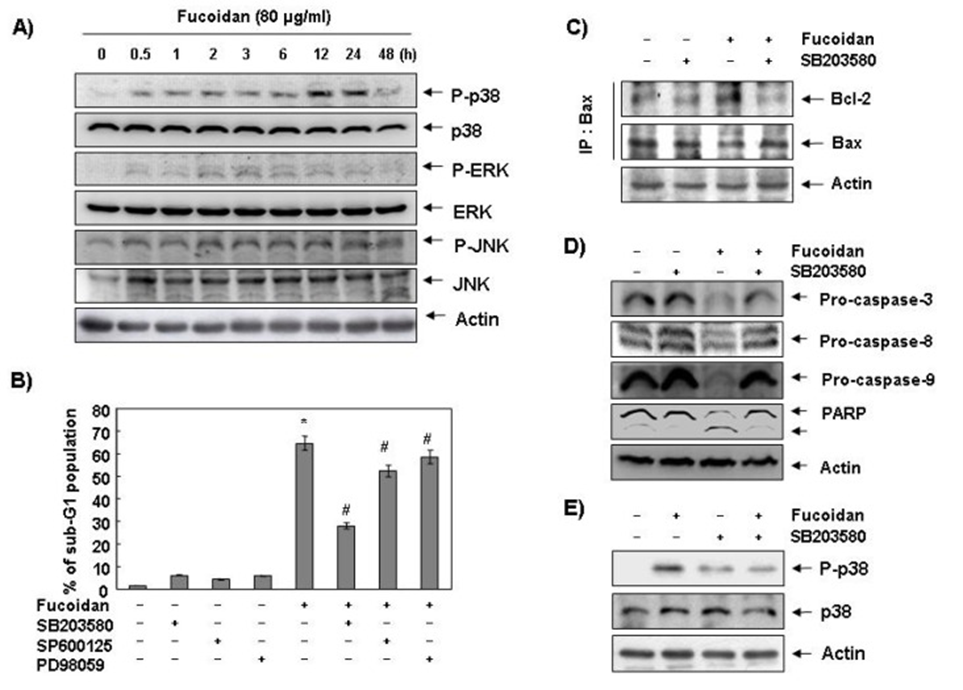
Following this, the study’s authors explored the impact of fucoidan on MAPK expression and activity. The goal was to discern whether these signaling pathways contributed to the apoptotic response that had been observed. As shown in Figure 6A, p38 MAPK protein phosphorylation levels significantly increased after 12 and 24 hours of fucoidan treatment, compared with ERK and JNK. Cells were pre-treated with MAPK inhibitors, and their sub-G1 DNA content was assessed using a flow cytometer to confirm the relationship between p38 MAPK activation and fucoidan-induced apoptosis. As shown in Figure 6B, pretreatment with SB203589 (a specific inhibitor of p38 MAPK) significantly reduced the fucoidan-induced increase in sub-G1 DNA content.
The absence of a significant effect when fucoidan treatment was preceded by PD98059 (a strong ERK inhibitor) or SP600125 (a strong JNK inhibitor) points to a direct correlation between fucoidan-induced apoptosis and p38 MAPK activation in U937 cells. To further elucidate the mechanism of fucoidan-induced p38 MAPK activation in U937 cells, we examined the binding of Bcl-2 and Bax and the expression levels of caspases. As shown in Figure 3C, pretreatment with a p38 MAPK-specific inhibitor significantly reduced the fucoidan-induced increase in Bcl-2 and Bax binding in U937 cells. The fucoidan-induced decline in procaspases 3, 8, and 9, as well as PARP cleavage and p38 MAPK phosphorylation, was restored by p38 MAPK inhibition (see Figures 3D and 3E). The data imply that fucoidan-stimulated p38 MAPK activation leads to caspase activation by facilitating Bax’s relocation from the cytoplasm to the mitochondria, culminating in programmed cell death.
In order to further elucidate the specific function that Bcl-2 plays in the process of apoptosis triggered by fucoidan, a selection process was implemented to identify and isolate G418-resistant clones exhibiting enhanced Bcl-2 expression, which were then employed in the ensuing experimental investigations. Results showed that Bcl-2 overexpression significantly protected U937 cells from fucoidan-induced sub-G1 cell formation and DNA fragmentation compared with U937/vector cells. Moreover, fucoidan therapy in U937 cells with elevated Bcl-2 levels completely reinstated the fucoidan-triggered drop in caspase-3, -8, and -9, as well as Bid activity and PARP cleavage.
The results suggest that enhanced expression of the anti-apoptotic protein Bcl-2 mitigates fucoidan-induced apoptosis, even though fucoidan treatment only marginally boosted the overall quantity of Bcl-2 protein. To find further evidence of the importance of Bcl-2 in fucoidan-induced apoptosis in U937 cells, we next examined the ability of HA14-1, a small molecule antagonist of Bcl-2, to reverse the effect of Bcl-2 on fucoidan-mediated apoptosis. When fucoidan and HA14-1 were administered together, they acted synergistically to significantly increase the proportion of sub-G1 cells from 8.5% to 33.8% in U937 cells that overexpressed Bcl-2 and had been treated with fucoidan. The involvement of Bcl-2 in fucoidan-induced apoptosis in U937 cells is significant, as indicated by these findings.
The study collectively shows that fucoidan significantly prompts apoptosis in human leukemia U937 cells. This occurs through the activation of caspases, a decrease in MMPs, the movement of Bax from the cytoplasm to mitochondria, and the release of cytochrome c from mitochondria to the cytoplasm, all mediated by the activation of p38 MAPK. This provides crucial mechanistic insight into how fucoidan triggers p38 MAPK-dependent apoptotic cell death in U937 cells.
Source: Mar Drugs. 2013 Jul 4;11(7):2347–2364. doi: 10.3390/md11072347
