Among the conventional methods for treating cancer are radiation therapy and chemotherapy; however, these treatments are commonly associated with considerable and debilitating side effects. Chemotherapy-induced peripheral neuropathy (CIPN) is a significant adverse effect of chemotherapy with restricted treatment choices. These chemo-drugs frequently irritate and damage sensory receptors, inducing CIPN and severely impacting treatment efficacy and patients’ quality of life. Over 70% of patients with CIPN exhibit clinical symptoms such as numbness, hypoesthesia, dysesthesia, and pain in their extremities. Thus, it needs a natural remedy that does not have side effects like fucoidan.
This blog post will discuss the study “Fucoidan reduces NET accumulation and alleviates chemotherapy-induced peripheral neuropathy via the gut–blood–DRG axis” by Rumeng Jia et al.
Research indicates that neutrophil extracellular traps (NETs) play a significant role in the development of chemotherapy-induced peripheral neuropathy (CIPN), with LPS/HMGB1 identified as a primary trigger for NET formation. They established a CIPN model using oxaliplatin (L-OHP). Mice were pretreated with fucoidan to examine the therapeutic effects.
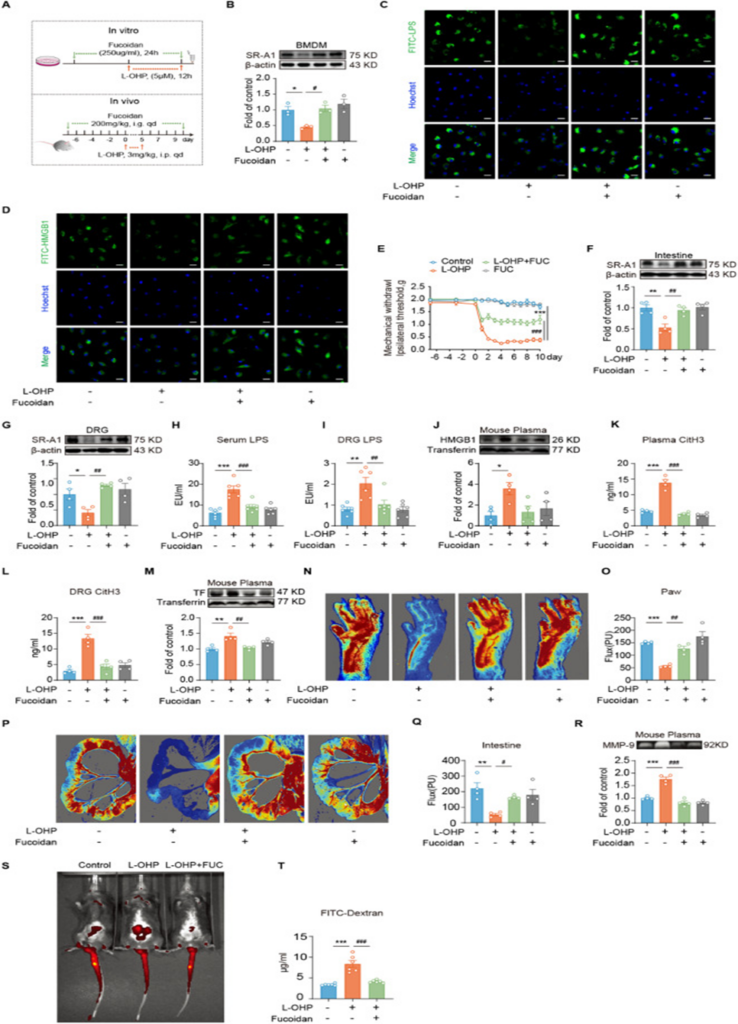
Scavenger receptor A1(SR-A1) receptor deficiency accelerates CIPN after chemotherapy, and SR-A1 upregulation is a key factor. As shown in Figure 1A, fucoidan upregulated SR-A1 receptor expression (see Figure 1B) and promoted the phagocytosis of FITC-LPS and FITC-HMGB1 by BMDMs (see Figures 1C and D). To further gauge the phagocytic activity of BMDMs, the researchers utilized fluorescent latex beads in our analysis. L-OHP treatment impaired the phagocytic function of BMDMs, as evidenced by a significantly reduced uptake of fluorescent latex beads compared to the control group. In contrast, fucoidan treatment effectively improved the phagocytic capacity of BMDMs.
In vivo, fucoidan attenuated L-OHP-induced mechanical hyperalgesia (see Figure 1E). We further examined the effect of fucoidan on SR-A1 expression in the intestine and DRG of mice. As shown in Figures 4F and G, fucoidan led to an increase in SR-A1 receptor expression within the intestine and DRG. The study also found that fucoidan decreased LPS accumulation in the plasma and DRG of CIPN mice (refer to Figures 4H and I) and reduced HMGB1 levels (see Figure 1J). This inhibited NET formation and reduced H3Cit levels in the blood and DRG (see Figures 4K and L). To further investigate the role of NETs, we performed immunohistochemical staining of DRG tissue sections using MPO (a marker for NETs) and Ly6g (a marker for neutrophils). The findings indicated that L-OHP treatment led to a significant rise in MPO-positive staining within the DRG tissue. This staining also showed substantial overlap with Ly6g, suggesting that L-OHP promotes the formation of NETs.
To gain further insight into NET formation, we also examined the colocalization of CitH3 and MPO in DRG tissue. The study observed that L-OHP treatment consistently elevated CitH3 and MPO, and their significant colocalization provided stronger evidence of increased NET formation post-L-OHP exposure. It’s important to note that fucoidan treatment successfully decreased the buildup of MPO and CitH3 in the DRG and prevented neutrophils from migrating to this region. This suggests that fucoidan may alleviate NET accumulation and exert potential therapeutic effects. The study observed that fucoidan brought about a decrease in TF expression (see Figure 1M), ameliorated circulatory dysfunction in the nerve terminals and intestine of mice (see Figures 1N-Q), and resulted in a reduction of MMP9 activity (see Figure 1R). The results point to a potential mechanism where fucoidan boosts LPS/HMGB1 clearance. By increasing SR-A1 in intestinal and DRG phagocytes, it may inhibit NET formation and mitigate circulatory problems, thereby offering relief from CIPN.
The increase in intestinal permeability caused by L-OHP was also prevented by fucoidan (see Figures 1S and T). After chemotherapy, the intestinal barrier in mice was disrupted, resulting in increased LPS leakage into the bloodstream and increased HMGB1 production, which further damaged the intestinal barrier and led to positive feedback damage. Fucoidan’s suppression of LPS/HMGB1 accumulation resulted in an indirect protective effect on the mouse intestine, breaking the cycle of malignant damage to the enteric nervous system.
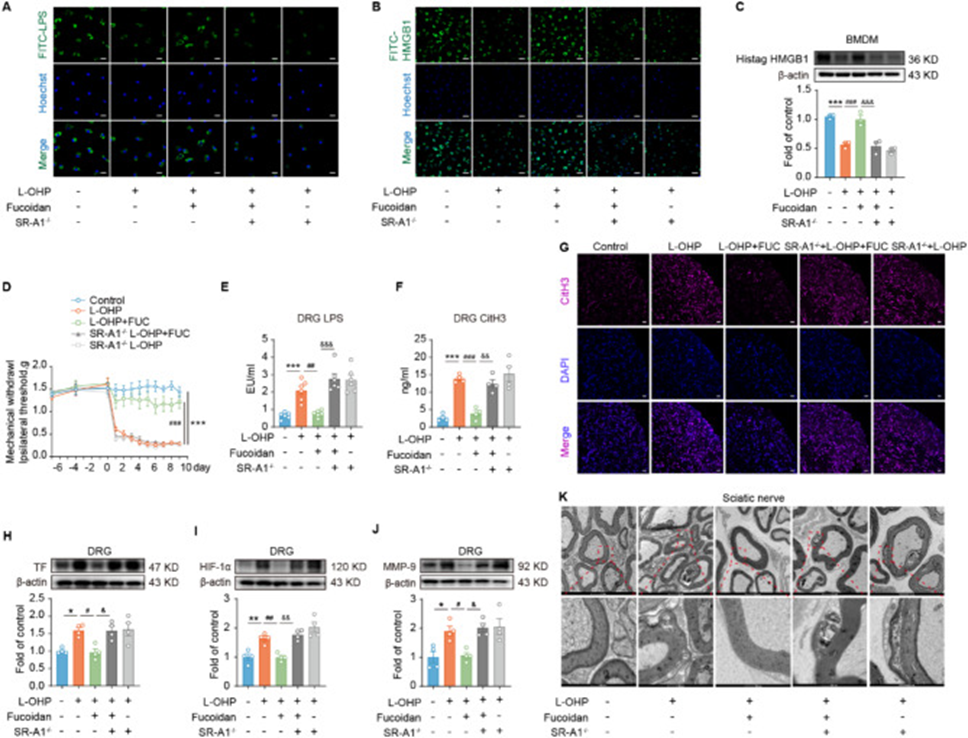
They further investigated the critical role of SR-A1 in the anti-CIPN effects of fucoidan by evaluating the therapeutic effects of fucoidan and related parameters using SR-A1-/- mice. Notably, we observed that the effects of fucoidan on LPS/HMGB1, MMP-9, and TF levels were abolished after SR-A1 knockout compared to the control group. In vitro results confirmed that fucoidan promoted the phagocytosis of FITC-LPS and FITC-HMGB1 by BMDMs, but this effect was abolished by SR-A1 knockout (see Figures 2A and B). The researchers swapped FITC-HMGB1 for His-tagged HMGB1 and then used Western blotting to analyze the His-tagged protein’s expression, aiming to understand how BMDMs phagocytose His-tagged HMGB1. Results showed that fucoidan increased the phagocytosis of His-tagged HMGB1 by BMDMs, which was also inhibited by SR-A1 knockout (see Figure 2C). Moreover, mice lacking SR-A1 exhibited a significant drop in their in vivo pain threshold (see Figure 2D), accompanied by a decline in associated indices and a build-up of LPS and H3Cit in the DRG (see Figure 2E-G).
Furthermore, TF expression and MMP-9 activity in the DRG were also increased (see Figure 2H and J). The assessment of the ischemic condition involved detecting HIF-1α levels, which revealed that fucoidan suppressed HIF-1α expression in the DRG of mice experiencing CIPN. However, after SR-A1 knockout, the hypoxic condition caused by high HIF-1α expression reappeared (see Figure 2I). Chemotherapy-treated CIPN mice showed a clear increase in sciatic nerve demyelination, but fucoidan attenuated this change. This change also disappeared in SR-A1 −/− mice (see Figure 2K). These results indicated that SR-A1 is crucial in preventing the buildup of LPS/HMGB1/NETs and lowering levels of MMP-9/TF/HIF-1α within the DRG of mice experiencing CIPN.
Furthermore, the study results showed that fucoidan suppressed the excessive accumulation of LPS/HMGB1/NETs in the blood induced by L-OHP, reduced plasma TF content and MMP-9 activity, and these changes were mediated by the SR-A1 receptor. Fucoidan can inhibit the formation of NETs and alleviate CIPN by promoting the phagocytic clearance of LPS/HMGB1. It was discovered that fucoidan can employ SR-A1 to engulf pre-formed NETs through phagocytosis.
They investigated the molecular mechanism by which fucoidan regulates the SR-A1 receptor. The “NEMETH_INFLAMMATORY_RESPONSE_LPS_UP” signaling pathway involved 87 genes, nine of which showed significantly differential expression. Among these nine genes, the Sqstm1 (p62) gene, located on chromosome 5, is a multifunctional protein that regulates the NF-κB signaling pathway. Furthermore, Sqstm1 can regulate SR-A1 expression. Subsequently, Sqstm1 was utilized to investigate the targets involved in fucoidan’s regulation of SR-A1. Research has shown that p62 assists Nrf2 escape into the nucleus through competitive binding with Keap1, ultimately regulating SR-A1 receptor expression. Research data showed a decrease in p62 expression in BMDMs subsequent to L-OHP stimulation, with an observed rise after fucoidan treatment.
Beyond that, nuclear Nrf2 protein levels were determined. Nuclear Nrf2 expression in BMDMs was decreased after L-OHP stimulation. Fucoidan increased Nrf2 protein levels. Co-IP results revealed that p62 binding to Keap1 decreased, and Nrf2 binding to Keap1 increased after L-OHP stimulation. Fucoidan treatment, however, led to a marked increase in Nrf2 nuclear translocation because p62 competed with Keap1 for binding. These results suggest that fucoidan promotes p62 binding to Keap1 and Nrf2, releasing Nrf2 into the nucleus and subsequently regulating SR-A1 transcription. Fucoidan’s effect on SR-A1, specifically its transcriptional upregulation and subsequent translation reversal, was confirmed with the Nrf2 inhibitor ML385. These results demonstrate that fucoidan acts on the SR-A1 receptor via the p62/Keap1/Nrf2 signaling pathway.
In addition to the previously mentioned findings, the sequencing analysis conducted on the microbial diversity demonstrated that the inclusion of fucoidan resulted in a noticeable augmentation of the gut microbiota’s diversity and abundance, accompanied by an increase in the Bacteroides/Firmicutes ratio. To summarize, fucoidan offers relief from CIPN by upregulating SR-A1 through the p62/Keap1/Nrf2 signaling pathway. This process enhances macrophage phagocytosis of HMGB1/LPS/NETs and preserves the balance of gut microbiota, potentially paving the way for novel approaches and treatments for CIPN.
Source: J Neuroinflammation. 2025 Apr 4;22:100. doi: 10.1186/s12974-025-03431-5
