Fucoidan, a sulfated polysaccharide, is predominantly extracted from the cell walls of brown algae. This substance primarily consists of sulfated L-fucose, with its composition of xylose, mannose, galactose, glucose, and uronic acid varying based on the specific type of brown algae. In addition to its anti-tumor activity, fucoidan is known to exhibit antioxidant, antibacterial, and immunomodulatory effects. Researchers are increasingly investigating fucoidan’s potential for treating cardiovascular illnesses, with a rising volume of research showing its effects on the heart and blood vessels. Fucoidan has been shown to mitigate atherosclerotic lesions through its anti-inflammatory and anticoagulant effects. Additionally, fucoidan has been demonstrated to inhibit neointimal formation and reduce vascular remodeling. This blog post introduces the research by Yuan Yao et al., “Fucoidan for cardiovascular application and the factors mediating its activities”. The objective is to offer a scientific review of fucoidan’s biological activities and functions pertinent to cardiovascular disease applications.
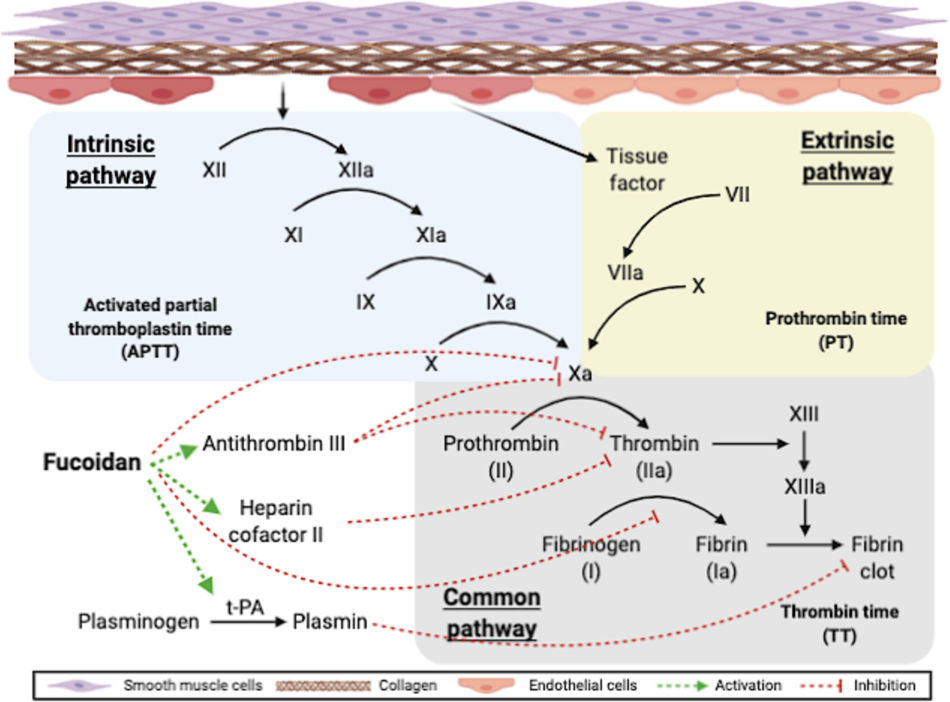
The coagulation cascade, illustrated in Figure 1, comprises three distinct pathways: the intrinsic, extrinsic, and common pathways. Activated partial thromboplastin time (APTT), prothrombin time (PT), and thrombin time (TT) are three important indicators of coagulation activity. APTT reflects coagulation activity in both the intrinsic and common pathways, PT indicates clot formation in the extrinsic pathway, and TT measures the thrombin-induced coagulation time in the common pathway. The first report of fucoidan exhibiting anticoagulant activity in vitro and in vivo was by Springer et al. The research indicated that giving fucoidan from Fucus vesiculosus through an IV led to a considerable extension of PT. Prolongation of APTT by fucoidan has been reported in many studies. Shang et al. investigated the anticoagulant activity of three types of fucoidan derived from sea cucumbers and showed that while APTT prolongation was significant, TT and PT were unaffected. The observation that fucoidan extends both APTT and PT indicates that it possesses anticoagulant properties affecting both the intrinsic and extrinsic pathways. The main targets of fucoidan have been identified as factor Xa and thrombin.
Factor Xa (FXa) is the activated form of Factor X and cleaves prothrombin to generate active thrombin. The interaction between fucoidan and FXa is considered important in the mechanism of fucoidan’s anticoagulant effect. Fucoidan is a potent inhibitor of FXa generation in the intrinsic coagulation pathway.
Fucoidan’s ability to enhance coagulation may be partly due to its suppression of tissue factor pathway inhibitor (TFPI).
The balance between the anticoagulant and procoagulant effects of fucoidan appears to be dependent on concentration and structure. Fucoidan demonstrated a procoagulant effect in a hemostatic thrombin generation assay with hemophilia patient plasma, starting at a concentration of 0.01 μg/mL and achieving peak activity around 1 μg/mL. However, at concentrations exceeding 10 μg/mL, the procoagulant effect disappeared. Zhang et al. revealed that the concentration profile showing both procoagulant and anticoagulant effects exhibits a bell-shaped curve.
Additionally, fucoidan has demonstrated antithrombotic properties because it inhibits thrombin. Intravenous administration of 1.8 mg/kg of fucoidan to rabbits significantly prolonged thrombin clotting time and suppressed thrombus formation with only a slight increase in bleeding volume. Thrombus formation in microvessels can lead to ischemia and even tissue infarction. Intravenous administration of fucoidan (25 mg/kg) has also been shown to effectively suppress thrombotic occlusion caused by endothelial denudation of arterioles and venules in vivo.
Fucoidan’s ability to inhibit thrombin primarily operates via pathways involving heparin cofactor II and antithrombin III. Heparin cofactor II and antithrombin III inactivate thrombin by forming complexes with it, blocking the active site of thrombin. Fucoidan has a high affinity for heparin cofactor II and strongly accelerates the heparin cofactor II-thrombin reaction rate. This activity of fucoidan is more effective than that of heparin. While fucoidan aids in thrombin inactivation through antithrombin III, it’s not as effective as heparin cofactor II.
Ustyuzhanina et al. found that achieving direct thrombin inhibition might require substantial amounts of fucoidan along with at least 16 sugar units.
In addition to its anticoagulant and antithrombotic effects, fucoidan also influences platelet adhesion, aggregation, and activation. Research indicates that fucoidan can suppress the aggregation of platelets stimulated by thrombin and adenosine diphosphate (ADP). Administering fucoidan intravenously at a rate of 0.5–1.0 mg/kg/h to baboons has been demonstrated to fully prevent platelet aggregation. Fucoidan has also been shown to inhibit platelet activation by reducing platelet aggregation.
Low molecular weight fucoidan fractions (15.2 kDa) have been shown to suppress platelet aggregation compared to high molecular weight fucoidan (170 kDa and 117 kDa). Other studies have also reported that high molecular weight fucoidan derived from Laminaria japonica induces platelet aggregation, while low molecular weight fucoidan inhibits thrombin-induced platelet aggregation.
The complement system is a surface-dependent and calcium-requiring reaction involved in innate immunity. Three pathways are involved in complement activation: the classical pathway, the mannan-binding lectin (MBL) pathway, and the alternative pathway. These three pathways lead to the formation of C3 convertase through different mechanisms.
Research suggests that the process of complement activation may contribute to the progression of atherosclerosis, thrombosis, and ischemic heart disease. Undesirable complement activation can occur in the blood or on the surface of cardiovascular implants via the classical and alternative pathways, potentially contributing to thrombus formation.
Fucoidan was first reported by Blondin et al. as a potent inhibitor of complement activation. Subsequently, Zvyagintseva et al. showed that fucoidans from Laminaria cichorioides, Saccharina japonica, and Fucus evanescens inhibited alternative complement activation by 50%, depending on the source. Fucoidan’s inhibitory effect on complement activation is mainly via the classical pathway, with a smaller impact on the alternative pathway. Fucoidan binds to C1q and inhibits the complete activation of C1 by interfering with the binding of the C1r2-C1s2 subunits. It has been hypothesized that fucoidan inhibits the cleavage of C4 by forming a complex with C4. Researchers later confirmed that fucoidan binds to C4 through nuclear magnetic resonance (NMR) analysis with fucoidan oligosaccharides. Furthermore, fucoidan directly inhibits the formation of C3 convertase. Fucoidan inhibits the formation of C4b2a by inhibiting the interaction between C4b and C2. Fucoidan also suppresses the production of C3bBb by reducing the number of C3b binding sites on factor B.
Selectins, which are glycoproteins, are vital for guiding leukocytes to inflammatory sites and damaged tissues, and for their subsequent buildup there. P-selectin is a cell adhesion molecule expressed by both endothelial cells and platelets. P-selectin has a high-affinity ligand, P-selectin glycoprotein ligand-1 (PSGL-1), which is expressed by leukocytes. When endothelial cells and platelets are activated, P-selectin translocates to the surface of activated endothelial cells and platelets, inducing leukocyte migration to the site of inflammation and contributing to thrombus formation. Fucoidan also acts as a ligand for P-selectin; its attachment to the carbohydrate recognition domain reduces the binding between P-selectin and leukocytes. L-selectin, expressed by most circulating leukocytes, recognizes endothelial mucin-like ligands and regulates leukocyte tethering, rolling, and activation. Fucoidan is also a ligand for L-selectin and can reduce leukocyte rolling and accumulation. By binding to selectins, fucoidan inhibits the adhesion of leukocytes, neutrophils, and platelets to the surface of damaged arteries. Across several animal models, fucoidan administration leads to a reduction in leukocyte rolling and neutrophil migration.
Fucoidan has also shown therapeutic effects in mitigating myocardial infarction by suppressing selectin-dependent platelet adhesion.
New research indicates that fucoidan not only hinders complement and selectins but also reduces the activity of various inflammatory enzymes. Oral administration of low-molecular-weight fucoidan derived from Saccharina japonica at a dose of 200 mg/kg/day has been shown to significantly reduce matrix metalloproteinase (MMP) levels, decrease the expression of interleukin-1β, tumor necrosis factor-α (TNF-α), and monocyte chemoattractant protein-1, and suppress the enlargement of abdominal aortic aneurysms (AAA). MMP-2 is involved in intimal thickening. Fucoidan derived from Fucus vesiculosus inhibits MMP-2 activity and reduces luminal narrowing in damaged blood vessels. In addition, the administration of fucoidan at a rate of 100 mg/kg/day has been found to diminish MMP activity by downregulating the activation of nuclear factor-κB and c-Jun N-terminal kinase (JNK).
Atherosclerosis, hypertension, and intimal thickening are all pathological conditions in which vascular smooth muscle cells (SMCs) are recognized to have a significant and vital role. In normal blood vessels, SMCs are in a quiescent state, differentiated and exhibiting contractility. However, when vascular endothelial cells (ECs) are damaged or dysfunctional, SMCs undergo a phenotypic change to a proliferative state and migrate from the media to the intima. This leads to intimal thickening, atherosclerotic plaque formation, and narrowing of the vascular lumen.
Fucoidan addition to cell culture media has demonstrated a significant suppression of SMC proliferation. McCaffrey et al. compared the SMC proliferation inhibitory effect of fucoidan to that of heparin and found that fucoidan exhibited a stronger inhibitory effect on SMC proliferation than heparin. Further investigations have shown that fucoidan curtails SMC proliferation by safeguarding transforming growth factor-β1 (TGF-β1) from degradation by plasmin and by amplifying TGF-β1 activity in SMCs. Fucoidan has also been reported to inhibit the phosphorylation and nuclear translocation of mitogen-activated protein kinase (MAPK) in SMCs. The antiproliferative effect of fucoidan depends on its structural characteristics.
In addition to inhibiting SMC proliferation, fucoidan also inhibits SMC migration. Low molecular weight fucoidan suppresses MMP-2 expression in SMCs and reduces SMC migration by 40 ± 3%. Recent studies using a mouse model of pulmonary arterial hypertension have shown that administration of fucoidan at a dose of 25 mg/kg inhibits the proliferation and migration of smooth muscle cells (SMCs) stimulated by hypoxia and growth factors. Fucoidan also significantly reduces methionine incorporation into fibronectin and increases thrombospondin synthesis. This may contribute to the inhibitory effect of fucoidan on SMC migration.
Fucoidan has been observed in multiple studies to inhibit the development of atherosclerotic lesions and prevent intimal thickening. Coating stents with fucoidan has been shown to suppress SMC proliferation and reduce the area of restenosis histopathologically. Xu et al. studied the mitigating effect of low-molecular-weight fucoidan on atherosclerosis in a mouse model. Fucoidan was found to ameliorate atherosclerotic lesions by diminishing SMC proliferation and migration, and by preventing macrophage formation and differentiation. A recent study developed a fucoidan-modified small-diameter vascular graft. Fucoidan modification significantly improved endothelial cell (EC) adhesion. The fucoidan-modified vascular grafts showed a lower stenosis rate than unmodified grafts, which is thought to be due to the inhibition of SMC proliferation and increased EC adhesion.
The research examined the impact of fucoidan on vascular cells’ physiological activity, coagulation, inflammation, and regulatory functions, with a focus on its cardiovascular system applications. The conclusions suggest fucoidan is a promising candidate for treating cardiovascular diseases.
Source:Carbohydr Polym. 2021 Jun 18;270:118347. doi: 10.1016/j.carbpol.2021.118347
