The chronic condition known as inflammatory bowel disease (IBD) is defined by the recurring nature of inflammation that affects the intestinal tract. It has two major subtypes: Crohn’s disease and ulcerative colitis (UC). UC is a particularly intractable disease characterized by high incidence, prolonged disease duration, and frequent recurrence. When the body doesn’t get enough dietary fiber, it can become more vulnerable to colitis. This might stem from a weakened intestinal barrier, the activation of the NF-κB pathway, and increased oxidative stress.
This blog post will discuss the study “Fucoidan alleviated colitis aggravated by fiber deficiency through protecting the gut barrier, suppressing the MAPK/NF-κB pathway, and modulating gut microbiota and metabolites” by Weiyun Zheng and colleagues. The research explored how mice experiencing colitis worsened by a lack of dietary fiber showed reduced symptoms when fed fiber supplemented with Undaria pinnatifida fucoidan (UPF).
First, mice were divided into three groups (n=9/group): normal group (NC), DSS + FD group (model), and DSS (dextran sulfate sodium) + FD (a fiber-deficient diet) + UPF (Undaria pinnatifida fucoidan) group (UPF). The NC group received standard food and water. In contrast, the other two groups were administered 2% DSS for five days, followed by water for three days, and then FD for eight days. The UPF group was orally administered 300mg/kg/day of UPF for 8 days.
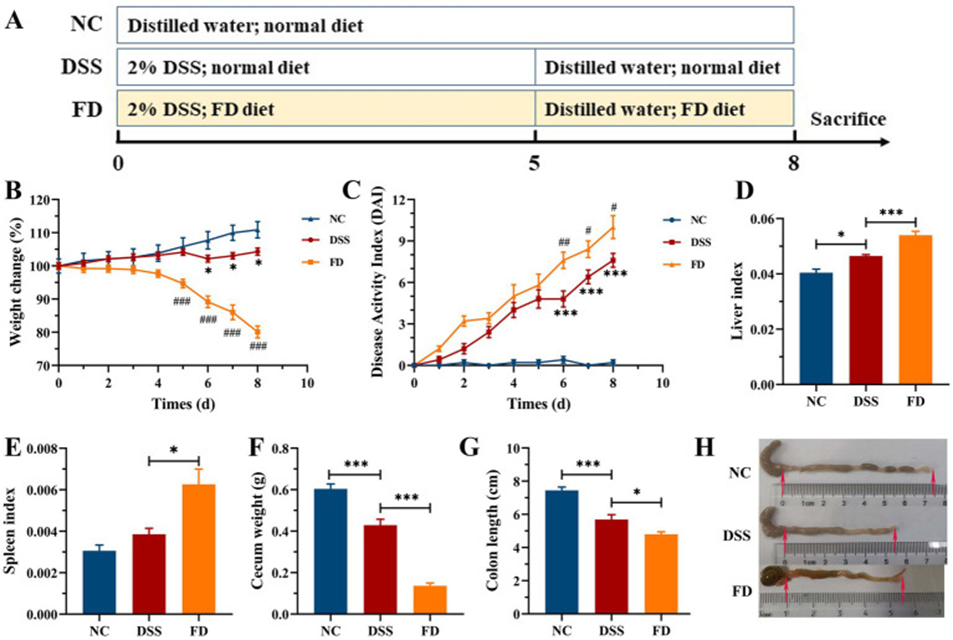
The effect of FD on colitis was first evaluated in mice administered 2% DSS (Figure 1). Mice in the model group showed clear symptoms of colitis, characterized by decreased body weight, cecum weight, and colon length, as well as increased liver index and DAI. FD exacerbated the aforementioned colitis symptoms, with a clear increase in splenic index. This suggests that FD may increase susceptibility to IBD in humans. This is similar to the high susceptibility of mice fed a fiber-free diet to DSS-induced colitis.
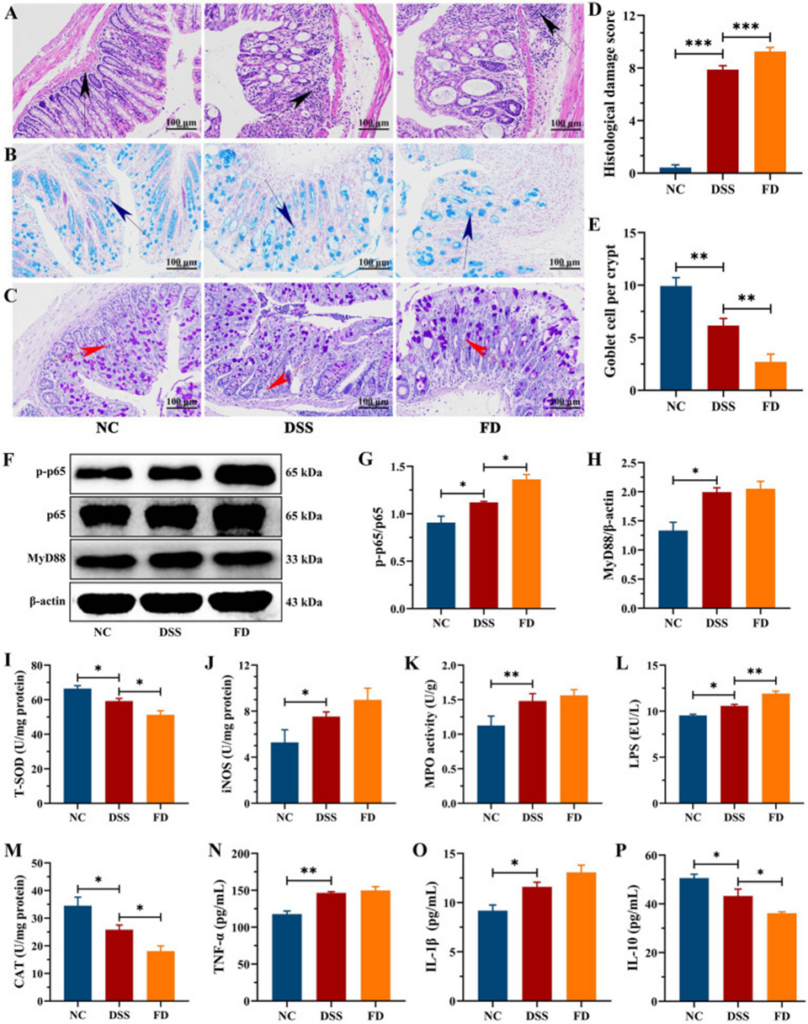
The HE staining showed substantial colon damage in the DSS group, with inflammatory cell infiltration, crypt loss, and mucosal erosion, resulting in a high histological score (as shown in Figure 2A and D). The mucus layer’s integrity was found to be compromised in the DSS group, as shown by AB and PAS staining. This was marked by a reduction in goblet cell count, a significant drop in glycoprotein density, and a thinner mucus layer (see Figure 2B, C, and E). The DSS group had higher levels of TNF-α and IL-1β and lower levels of IL-10 than the NC group (Figure 2N–P). This indicates that DSS treatment caused colonic inflammation. FD exacerbates colitis, as evidenced by increased crypt loss, inflammatory infiltration, and IL-1β levels, but also decreased IL-10 levels and mucus layer thickness. These findings point to a connection between FD and the beginning and worsening of inflammatory bowel disease (IBD).
The recurrence and progression of IBD are critically dependent on the delicate interplay between oxidants and antioxidants, and excessive ROS production disrupts this vital balance. The contents of iNOS and MPO were significantly higher in the DSS group than in the NC group, and the activities of CAT and T-SOD were reduced (Figures 2I–K, M). CAT and T-SOD are key components of the antioxidant defense network and play a crucial role in scavenging ROS. By creating reactive oxidants such as hypochlorous acid, MPO restricts the availability of nitric oxide (NO), potentially causing endothelial dysfunction. Excessive production of NO, catalyzed by iNOS, can cause cytotoxicity. The study found that FD reduced CAT and T-SOD levels while elevating iNOS levels, which consequently worsened inflammation in the colon.
Additionally, the model group experienced substantial colitis symptoms, including a decline in body weight gain, colon length, cecum weight, spleen index, and liver index, as well as elevated DAI, inflammatory infiltration, and tissue damage. UPF increased body weight gain and cecum weight in mice and decreased DAI, liver index, and spleen index. UPF alleviated the inflammatory response, as evidenced by reduced inflammatory infiltration, crypt loss, colonic edema, and histological scores. The findings align with those of fucoidan, which was observed to lengthen the colon and diminish the spleen index, DAI, and colonic inflammation in mice. This suggests that UPF can function as a prebiotic dietary fiber for maintaining intestinal health.
In the model group, the levels of the proinflammatory cytokines TNF-α and IL-1β were significantly higher than in the NC group, while IL-10 levels were significantly reduced. UPF did not affect IL-10 levels, but reduced IL-1β and TNF-α levels, demonstrating anti-inflammatory activity. To explore the potential mechanisms, we evaluated the effects of UPF on key proteins in the MAPK/NF-κB pathway and oxidative stress-related markers. UPF significantly reduced the levels of p-IκBα/IκBα, TLR4, and MyD88 in DSS-treated mice. This suggests that UPF can alleviate colitis by inhibiting the activation of the NF-κB pathway. UPF improved the imbalance between oxidative stress and antioxidant defense by increasing CAT levels, which decomposes H2O2 into water and oxygen, enhancing T-SOD activity, which neutralizes ROS, and reducing MPO levels. This indicates that UPF may help decrease inflammation in the colon through the reduction of oxidative stress.
In addition to its other benefits, UPF contributes to the alleviation of colonic inflammation by actively lowering the levels of intestinal LPS, a process that subsequently diminishes the infiltration of inflammatory factors into the colon. In the model group, the ratios of p-JNK/JNK, p-p38/p38, and p-ERK/ERK were significantly increased, suggesting activation of the MAPK pathway. UPF treatment reduced the levels of p-p38/p38, p-JNKs/JNKs, and p-ERKs/ERKs, suggesting that UPF can suppress inflammatory responses by inhibiting signaling of this pathway. The study’s outcomes are consistent with Mesona chinensis polysaccharides’ ability to lessen colonic inflammation by downregulating the MAPK/NF-κB pathway.
The model group exhibited a compromised mucus layer, marked by a reduction in goblet cells, the disappearance of crypts, and a diminished mucus layer thickness. However, UPF helped prevent the loss of crypts, the decrease in goblet cells, and the reduction in glycoproteins, thus maintaining the mucus layer’s structural integrity. The protein complex known as tight junctions (TJs) includes multiple claudin and occludin members, and the cytoplasmic protein ZO-1, which binds to the actin cytoskeleton.
In this study, the distribution and expression of TJs and MUC2 in the mouse colon were examined by IHC. Brown and light brown sections were considered positive. Claudin-1, occludin, and ZO-1 were clearly reduced in the model group compared with the NC group. Disruption of intestinal epithelial junctions leads to increased epithelial permeability, facilitating the translocation of toxic substances such as LPS. UPF increased the expression levels of ZO-1, claudin-1, and occludin, maintained the integrity of intestinal epithelial tight junctions, and reduced the penetration of toxic substances, suggesting that β-glucan reduced colonic inflammation in mice through its effects on the mucus layer and intestinal epithelial tight junctions.
There was no difference in the Shannon index between the model and UPF groups. The Chao1 index decreased in the model group (p>0.05) but increased in the UPF group, suggesting that UPF can enhance the richness of the microbiota. PCoA and UPGMA clearly distinguished the NC and model groups, yet no difference was observed between the UPF and model groups. A higher level of microbial diversity was linked to better outcomes for UC patients undergoing fecal microbiota transplantation. This indicates that UPF could be advantageous for gut health by increasing the diversity of gut bacteria, potentially warding off the development of colitis.
The model group had significantly higher levels of Bacteroidetes and Proteobacteria, while Firmicutes (p>0.05) and Actinobacteria were lower compared to the NC group. In contrast, the UPF group had decreased levels of Proteobacteria and Bacteroidetes, while Firmicutes levels increased. This indicates that UPF could help manage IBD by lowering levels of Bacteroidetes and Proteobacteria while boosting Firmicutes.
The study utilized HPLC/MS to determine the alterations in microbiota metabolites due to UPF consumption. Principal Component Analysis (PCA) utilizing both ESI+ and ESI− modes effectively distinguished between the NC and model groups. This separation demonstrates that the metabolic profile of the microbiota undergoes changes when colitis develops. The microbiota metabolite profile was regulated by UPF, and a clear separation was observed between the UPF and NC groups. Furthermore, OPLS-DA showed clear differentiation among the NC, UPF, and model groups. The findings imply that UPF can modify, yet not restore, the changes present in the microbial metabolite profile.
These findings suggest that FD could increase a host’s susceptibility to IBD by undermining the intestinal barrier’s functionality and disrupting the gut’s microbial equilibrium. UPF attenuated FD-exacerbated colitis, which was associated with maintaining the integrity of the intestinal barrier and inhibiting oxidative stress and the MAPK/NF-κB pathway. It’s believed that the core process involves altering the gut’s microbial community and its byproducts, specifically by boosting Bifidobacteria, spermidine, and carnitine, while reducing Bacteroides and deoxycholic acid. Based on this evidence, it is proposed that UPF might serve as a prebiotic dietary fiber, offering benefits that contribute to the promotion of human health.
Source: Front Nutr. 2025 Jan 24;11:1462584. doi: 10.3389/fnut.2024.1462584
