The increasing prevalence of nonalcoholic fatty liver disease (NAFLD) is a significant trend in chronic liver disease, with an estimated 25% of people worldwide suffering from it. It is noteworthy that non-alcoholic fatty liver disease (NAFLD), and more specifically its histological manifestation termed nonalcoholic steatohepatitis (NASH), carries the risk of advancing to severe liver disease, including the stages of fibrosis, cirrhosis, and ultimately the development of hepatocellular carcinoma. Despite the current lack of effective NAFLD therapies, approaches aimed at diminishing hepatic fat buildup, oxidative stress, and inflammation show promise as future treatments.
This blog post will feature the study “Fucoidan and Fucoxanthin Attenuate Hepatic Steatosis and Inflammation of NAFLD through Modulation of Leptin/Adiponectin Axis,” authored by Ping-Hsiao Shih et al. The study explored the joint impact of LMF-HSFx, a blend of low-molecular-weight fucoidan and highly stable fucoxanthin, as a potential treatment for NAFLD.
The initial step to investigate the protective effect of LMF-HSFx on NAFLD-induced hepatic lipotoxicity involved randomly assigning 42 patients to either the LMF-HSFx or placebo group. NAFLD patients received 24 weeks of oral LMF-HSFx or placebo treatment, and serum biochemical parameters were investigated. After 6 months, the LMF-HSFx group showed significant reductions in serum alanine aminotransferase (ALT) and aspartate aminotransferase (AST) relative to baseline. After 6 months, the LMF-HSFx group also showed significant reductions in total cholesterol (TC) and triglycerides (TG) relative to baseline. Evidence suggests that LMF-HSFx supplementation effectively lessens hepatic lipotoxicity in individuals suffering from NAFLD.
As depicted in Figure 1A, a significant decrease in CAP values (relative to baseline percentages) was observed in the LMF-HSFx group after six months of LMF-HSFx treatment. Interestingly, the stiffness values (% relative to baseline) were also considerably reduced (see Figure 1B). These data suggest that LMF-HSFx reduces hepatic steatosis and fibrosis in patients with NAFLD.
The study examined shifts in inflammatory cytokines, namely IL-6 and IFN-γ, and adipokines, including adiponectin and leptin, within NAFLD patients who were administered LMF-HSFx or a placebo. Over a period of 3 to 6 months, notable declines in IL-6 and IFN-γ were evident within the LMF-HSFx group.
Adiponectin has been linked to impaired mitochondrial function and insulin resistance in diabetes, according to research. Another major adipokine is leptin, whose actions include promoting fatty acid oxidation, glucose reduction, and weight and fat loss. Regarding adipokines, both adiponectin and leptin were significantly increased relative to baseline.
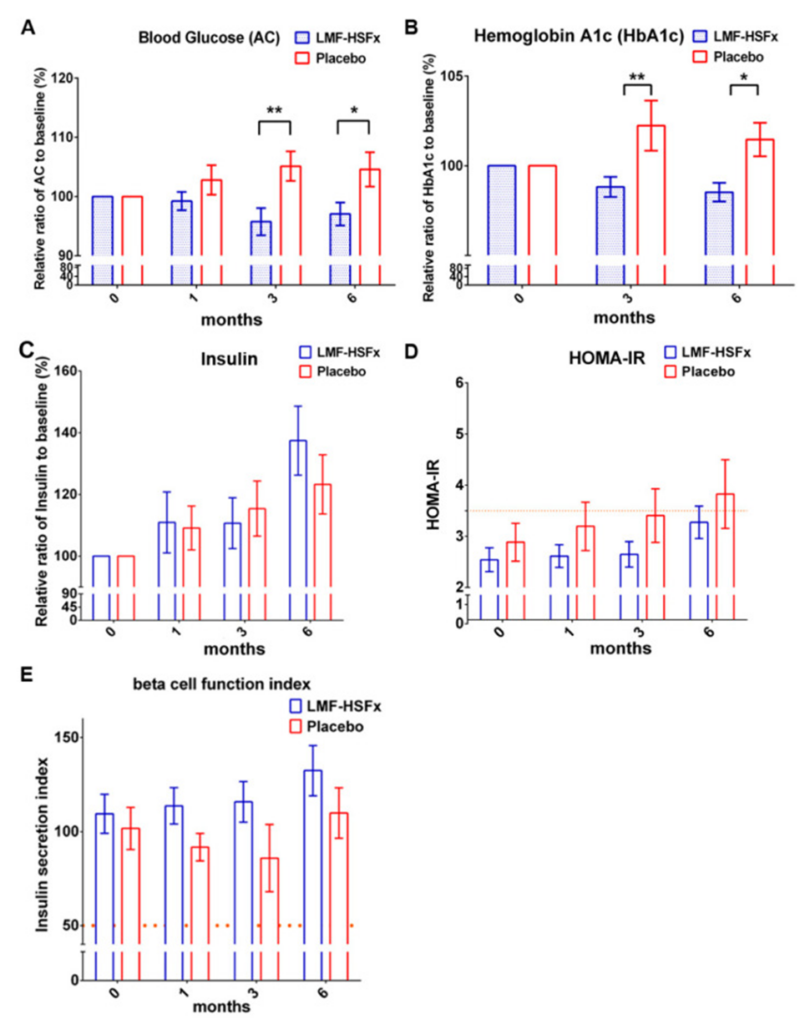
They next investigated whether LMF-HSFx attenuates insulin resistance in NAFLD. The data showed that after 6 months of LMF-HSFx treatment, fasting plasma glucose (AC) and hemoglobin A1c (HbA1c) levels significantly decreased (see Figure 5A, B). The LMF-HSFx group demonstrated a rise in relative insulin levels, expressed as a percentage compared to the baseline, as illustrated in the figure. In addition to their other analyses, the researchers calculated the homeostasis model assessment of insulin resistance (HOMA-IR) and the insulin secretion index, which are valuable metrics for understanding both insulin resistance and the capacity of the beta cells. Compared with the placebo group, the LMF-HSFx group had a lower HOMA-IR (see Figure 5D), but a higher insulin secretion index (see Figure 5E). This suggests that LMF-HSFx may attenuate insulin resistance and promote β-cell repair in NAFLD.
Subsequently, the researchers examined how LMF-HSFx impacted liver cell damage caused by fat in mice. Mice fed an HFD were orally administered LMF-HSFx (400 mg/kg/BW/day) for 16 weeks. LMF-HSFx significantly suppressed lipid accumulation in the liver. The amount and size of lipid droplets in liver tissue were analyzed, and the data suggested that LMF-HSFx supplementation significantly reduced lipid droplet volume in the liver of HFD-fed mice. Fasting blood glucose levels were markedly higher in HFD mice than in ND mice. However, when mice on a high-fat diet (HFD) were given 400 mg/kg/BW/day of LMF-HSFx, their serum glucose levels were substantially lowered. The TG content of HFD mice treated with LMF-HSFx was also lower than that of the HFD group. It was also observed that HFD mice had significantly elevated serum AST and ALT levels compared to ND mice, suggesting that LMF-HSFx played a significant role in reducing HFD-induced lipotoxicity in the liver. Next, they evaluated the effect of LMF-HSFx on HFD-induced adipogenesis dysregulation in mice. In adipose and pre-brown adipose tissues, LMF-HSFx significantly upregulated the adiponectin expression genes adipoq and adig, as well as the leptin expression gene lep.
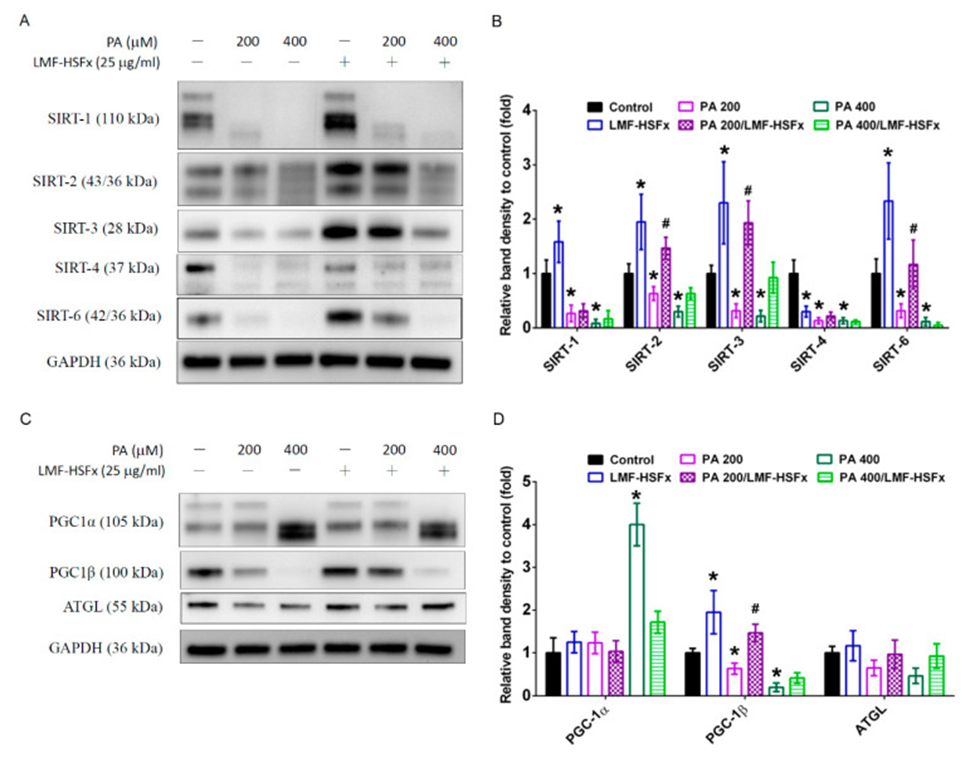
The expression of SIRT family proteins was diminished in HepaRG cells following PA-induced hepatic lipotoxicity, according to Figures 3A and 3B. This expression is regulated by the leptin-adiponectin axis. Additionally, PA treatment markedly triggered SIRT cleavage or reduced expression proportionally with the dose. In contrast, LMF-HSFx effectively reversed the PA-induced breakdown of SIRT2, 3, and 6 (see Figure 3A). SIRT1, SIRT3, and SIRT6 levels were significantly increased in the LMF-HSFx group compared with the control group (see Figure 3B). Furthermore, PA-treated hepatocytes showed a dose-dependent increase in PGC-1α but a decrease in PGC-1β, and LMF-HSFx significantly restored these effects (see Figure 3C, D). Consequently, the level of adipose triglyceride lipase (ATGL), a downstream lipid metabolism-related protein, was also significantly restored by treatment with 25 μg/mL of LMF-HSFx (see Figure 3C, D).
The study examined the ability of LMF-HSFx to prevent cell death in HepaRG cells exposed to PA. PA at 200 and 400 μM significantly increased the percentage of cells undergoing early apoptosis compared with the control group (3 ± 1.8%). LMF-HSFx rescued PA-induced cell death and DNA fragmentation in HepaRG cells. LMF-HSFx also rescued PA-induced cell cycle arrest and reduced PA-induced caspase 3 activation in HepaRG cells. Moreover, researchers examined the mitochondrial membrane potential in HepaRG cells subjected to treatment with or without 25 μg/mL LMF-HSFx. JC-1 staining assay confirmed that PA reduced mitochondrial potential in a dose-dependent manner. LMF-HSFx significantly improved mitochondrial integrity compared with the control group.
The findings collectively indicated that MF-HSFx led to improvements in hepatic steatosis, inflammation, fibrosis, and insulin resistance among individuals diagnosed with NAFLD. LMF-HSFx has the potential to impact the leptin-adiponectin system in adipocytes and hepatocytes, leading to regulation of lipid and glycogen metabolism, a reduction in insulin resistance, and the suppression of NAFLD.

Source: Mar Drugs. 2021 Mar 12;19(3):148. doi: 10.3390/md19030148
